Before I explain to you about the entropy, let me ask you a question.
What do you do in your classroom in the presence of a strict teacher?
You sit silently and disciplined in the classroom. Right?

But what do you do during a recess time?

This is the exact situation which happens during the recess time.
Now what is the Definition of Entropy in thermodynamics?
Here is a definition:
The measurement of randomness of the system is known as Entropy.
Or
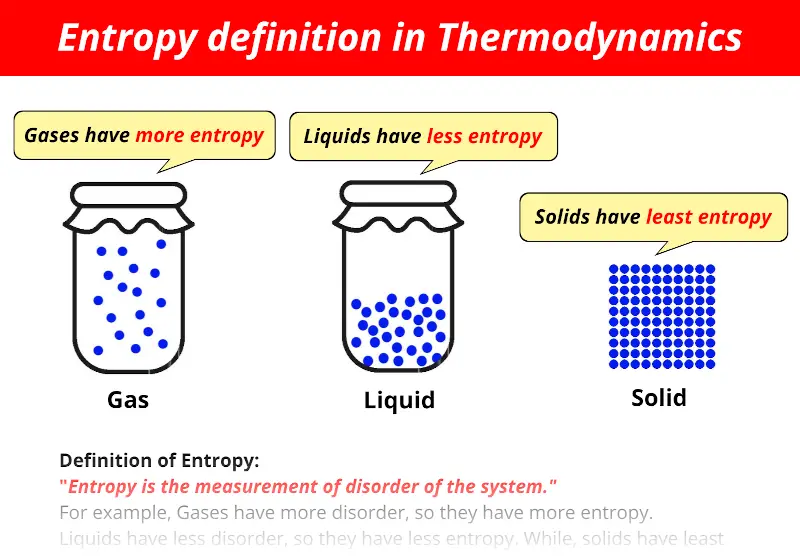
Entropy is the measurement of disorder of the system.
I know you have not understood anything in this definition. (Hahaha)
Don’t worry, I’ll explain this to you in a simple way.
For understanding the definition of entropy, you should know what is randomness or disorder?
Randomness: The movement of molecules is known as randomness.
Let me explain this with the above example.

Imagine that there is a strict teacher in a classroom. What will be the situation of all the students?
You can see that all the students are sitting in a proper disciplined manner and they are also sitting at regular intervals. These students are not moving at all. That means there is no disorder or no randomness in the classroom.

Now this is the situation during a recess time.
What did you notice here?
You can see that the students are moving here and there during a recess time.
That means they show disorder or randomness.
Thus we can say that the randomness or disorder is more.
This is a simple concept of randomness or disorder.
Now, let me explain this entire concept of randomness in solids, liquids and gases.
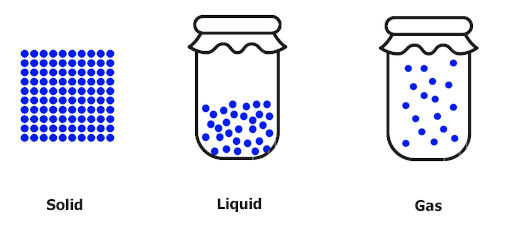
What about the movement of molecules in solids, liquids and gases.
You can see in the image that the solids show proper arrangements of molecules. They do not show any molecular movement. Thus solids have very very less disorder or very less randomness.
Now what about the liquids?
You can see in the above image that liquids show more movement of molecules as compared to solids. Thus the randomness of liquids is more as compared to solids.
In gases, you can see that the entire container is filled with the gas (in other words, gas occupies the entire space of the container).
Gas molecules show maximum movement and thus we can say that it has the maximum randomness or maximum disorder.
In short, we can say that;
The movement of molecules is known as randomness or disorder.
Now let’s move to entropy.
Contents
What is Entropy?
Few seconds ago, we discussed about the randomness. Now, entropy is nothing but a measurement of this randomness in any system.
Now, let me give you a simple definition of entropy.
Definition:
“The measurement of randomness of the system is known as Entropy.”
Or
“Entropy is the measurement of disorder of the system.”
It’s simple, it is just a measurement of how much randomly the molecules are moving in a system.
- In solids, the molecules are properly arranged, which means it has less randomness, so the entropy of solids is least.
- In gases, the molecules move very fast throughout the container. It has more randomness which means it has more entropy.
- The entropy of liquids lies in between the solids and liquids.
The entropy is denoted by the alphabet “S”.
The fact: We can not measure the exact entropy of any system. But we can only measure the change in the entropy (∆S) of the system.
The formula for change in entropy is given by the equation;
∆S = ∆Q/T
The unit of entropy is J/K.
Entropy in thermodynamics laws
Entropy in second law of thermodynamics
Here is the entropy statement of second law of thermodynamics.
“In all the spontaneous processes, the entropy of the universe increases.”
Second law of thermodynamics equation (formula) can be stated as below;


This entropy equation is very important as it tells us whether the process will occur on it’s own or not.
But what is a spontaneous process?
Spontaneous process is a process which occurs on it’s own without any external help.
We can easily find out whether the process is spontaneous or not by simply calculating the entropy change of the universe (∆Suniverse)
I would like to give you a few examples of spontaneous processes in which the entropy of the universe increases.
Examples of spontaneous processes
1). Melting if ice is automatic

2). Cooling of hot coffee is automatic

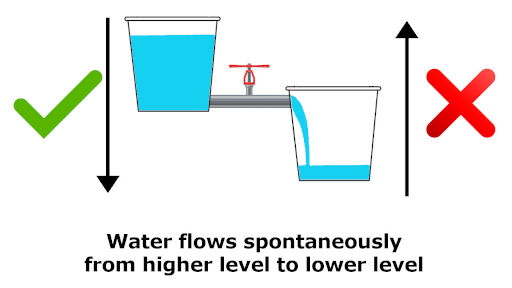
3). Water always flows from higher level to lower level
4). Gas occupies entire volume of the container on its own
5). Object falls on the ground on its own

6). Air leaks from the balloon on its own

These processes occur on their own. So they are known as spontaneous processes. For all these processes, the entropy of the universe always increases according to the entropy statement of the second law of thermodynamics.
For more information about the entropy statement of second law of thermodynamics, you can visit this article – “Everything about second law of thermodynamics“
Entropy in third law of thermodynamics
First of all, let me tell you how entropy is related to the third law of thermodynamics.
Let us see the statement of 3rd law of thermodynamics.
“The value of entropy of a completely pure crystalline substance is zero at absolute zero temperature”
Means if we have a pure crystalline solid substance and if it’s temperature is absolute zero (0 K), then it’s entropy will be zero.
Let me explain this to you with a simple image.
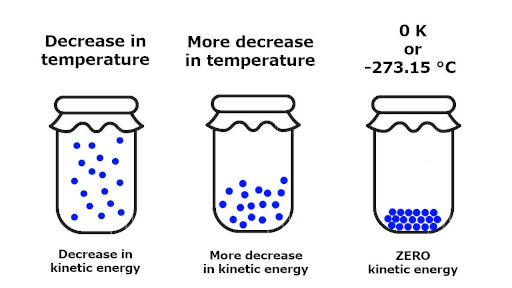
You can see that the gas is filled inside the container.
If we decrease the temperature of the system, the kinetic energy of the gas molecules also decreases.
Again if we keep on decreasing the temperature of the system, then there will be more decrease in kinetic energy of the molecules.
Now here is the main part.
If we keep on reducing the temperature and if we reach the temperature of 0 K (or -273.15 °C), then all the molecular motion will stop. The kinetic energy of the molecules will become zero.
This indicates that there is no disorder or randomness in the system.
Thus we can say that the entropy of such substances at absolute zero temperature is zero.
For more information about third law of thermodynamics, you can check out this article – Everything about 3rd law of thermodynamics.
I hope you have clearly understood the concept and definition of entropy in thermodynamics.
If you have any queries, feel free to comment below in the comments section.
Also let me know, which is the best example you enjoyed the most.
Important Guides for you
Also read:
- What is thermodynamics? (35+ topics with definitions and examples)
- Zeroth law of thermodynamics
- What is First law of thermodynamics?
- First law of thermodynamics definition/statement (In simple way)
- Examples of First Law of Thermodynamics / Law of conservation of Energy
- First law of thermodynamics equation (A practical explanation)
- Limitations of First Law of Thermodynamics
- What is second law of thermodynamics? [8+ Best examples to remember the law]
- Examples of Second Law of Thermodynamics (8+ best examples)
- Second Law of Thermodynamics Definition/Statement (Next level explanation)
- Second Law of Thermodynamics Equation [Practical explanation]
- What is third law of thermodynamics? (in simple terms)
- Laws of thermodynamics
- What is Carnot Cycle in Thermodynamics?
- Heat capacity vs specific heat in thermodynamics
- What is Thermodynamic Equilibrium? (With Best Example)
- Thermodynamic Process (With Examples)
- What is Thermodynamic System? – Open, Closed & Isolated system (With Examples)
