
Yes, really really simple !!
Let me ask you a question.

If you keep this hot coffee in your room, then it will cool down automatically. Right??

Now just ask yourself, will this coffee again become hot on its own?
The answer is big NO.
It will not absorb the heat from the surrounding on its own.
Well, second law of thermodynamics is all about this only.
Don’t worry, I’ll explain to you the statement/definition of second law of thermodynamics in a very simple way using real life examples.
Contents
How many statements are there for 2nd law?
The simple answer is THREE.
There are 3 statements for second law of thermodynamics.
1) Second law of thermodynamics for heat engine (Kelvin Planck’s statement)
2) Second law of thermodynamics for heat pump/refrigerator (Clausius’s statement)
3) Second law of thermodynamics based on entropy
You might have some questions regarding “What is the second law of thermodynamics in physics?” Or “What is the second law of thermodynamics in chemistry?”
Look, the actual meaning of the laws remains same only, whether it is in physics, chemistry or biology.
But many students get confused because this second law comes under physics as well as chemistry.
See, the statement for 2nd law was given by many scientists/physicists in their own way.
The Kelvin Planck’s statement and Clausius’s statement are related to heat engine and heat pump respectively.
So based on these applications, the Kelvin Planck’s statement and Clausius’s statement has been included in physics, and the Entropy statement of the second law has been included in chemistry.
Now, let me discuss with you all these three statements.
1) Second law of thermodynamics for heat engine (Kelvin Planck’s statement)
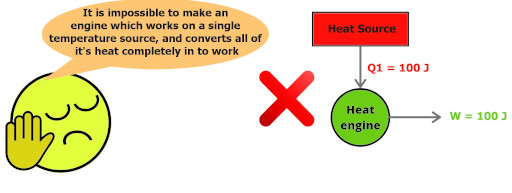
This single image will make you understand the entire statement very easily.
Kelvin Planck’s statement:
“It is impossible to construct a device (operating in a cycle) which works on a single heat source and converts all of its heat completely in to work”
I know it’s tough to understand such statements given by great scientists and physicists.
But I’m here to simplify this. Don’t worry.
Kelvin Planck’s statement simply wants to say that – “A heat engine must exchange the heat with at least two thermal reservoirs, one at higher temperature and other at lower temperature, then only the engine will operate.”
Or
“Perfect engine with 100% efficiency is impossible”
Let me clarify this with an example.
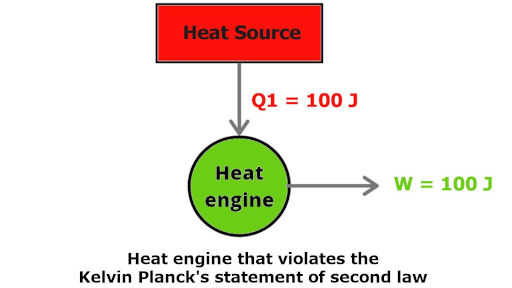
This image shows that the heat engine is continuously producing work by absorbing the heat energy from the heat source.
100 kJ of work heat is absorbed and exactly 100 kJ of work is done by that engine.
According to first law of thermodynamics, heat energy is converted into work energy (Energy is converted from one form to another)
If we look at this phenomenon from the first law perspective, then it is true.
Good…
But, according to Kelvin Planck’s statement, this is impossible.
It is impossible to have 100% efficiency of any heat engine.
Also the heat engine is not having any lower temperature reservoir to absorb the heat.
So it is impossible to have such an engine working on only a single reservoir.
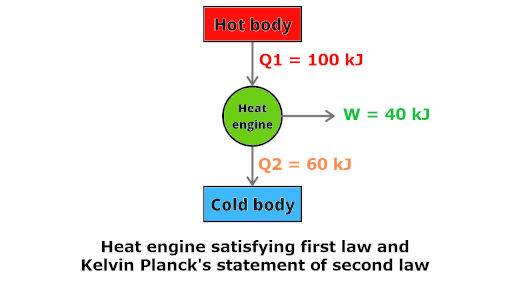
Now this type of engine is possible according to Kelvin Planck’s statement of second law of thermodynamics.
Because you can clearly see that there are two thermal reservoirs, one at higher temperature and other at lower temperature.
This engine satisfies the 1st law of thermodynamics as well as Kelvin Planck’s statement of second law.
2) Second law of thermodynamics for heat pump (Clausius’s statement)
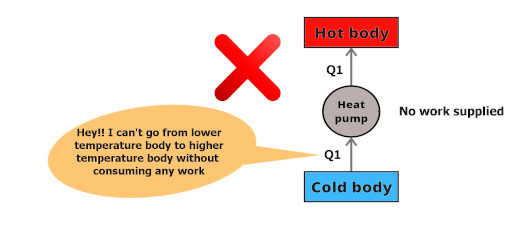
Here is a statement;
Clausius’s statement:
“It is impossible to construct a device (operating in a cycle) that can transfer heat from cold body to the hot body without absorbing any work.”
Or
“Heat can not itself flow from colder body to a hotter body.”
Again, I want to say that it is difficult to understand such statements given by great scientists.
Let me simplify this.
You can see that the heat Q1 is at a lower temperature body and it is saying that it will not travel to a higher temperature body without absorbing any work.
Let me explain this in detail.
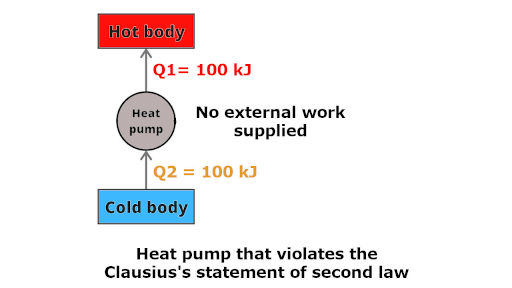
This is a heat pump in which 100 kJ of heat is extracted from the lower temperature body and the same 100 kJ of heat is transferred to the higher temperature body without supplying any work to it.
This phenomenon is possible according to the first law of thermodynamics (energy can be transferred from one body to another)
But according to the Clausius’s statement, this is impossible.
Heat can not itself flow from lower temperature body to higher temperature body.
Thus this heat pump violates the Clausius’s statement of second law.
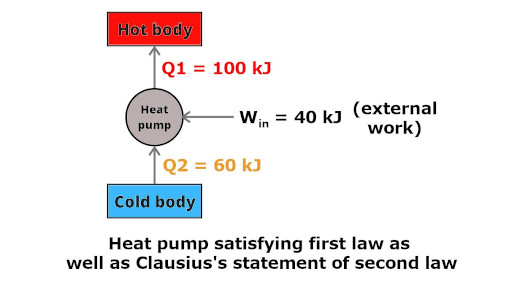
Now see, this type of heat pump is possible according to Clausius’s statement of second law because it consumes some external work (40 kJ).
This heat pump obeys first law as well as second law of thermodynamics.
3) Second law of thermodynamics based on entropy
“In all the spontaneous processes, the entropy of the universe increases.”
This can be written in equation form as;


Now you might have questions in your mind that…
What is the spontaneous process?
What is entropy?
Spontaneous processes are those processes which occur on it’s own.
(For example,
- Gas spreads in the entire room on its own,
- Hot coffee cools down automatically on its own,
- Ice kept on the table will melt on its own, etc.
For more examples of spontaneous process, kindly refer this article – “Examples of second law of thermodynamics“)
Now, what is entropy?
You all know about solids, liquids and gases.
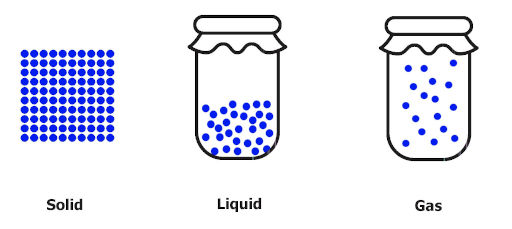
Solids have closed arrangements of molecules.
Liquids shows more movement of molecules compared to that of solids, and
Gases show maximum movement of molecules as shown in the above picture.
Now, this movement of molecules is known as disorder or randomness.
And measurement of this disorder is known as entropy.
Thus entropy can be defined as follows.
Entropy is the measurement of disorder of the system.
Thus the entropy statement of second law of thermodynamics simply says that – “If the process is occurring on its own, then the disorder of the universe always increases.”
If you want the mathematical proof for this above entropy statement of second law, then visit this article: “Second law of thermodynamics with examples“
In this article I have given a mathematical proof that the entropy of the universe always increases for all spontaneous processes.
Now, let me summarize all these three statements/definition of second law of thermodynamics for your better revision.
Summary
1) Second law of thermodynamics for heat engine (Kelvin Planck’s statement)
“It is impossible to construct a device (operating in a cycle) which works on a single heat source and converts all of its heat completely in to work”
2) Second law of thermodynamics for heat pump/refrigerator (Clausius’s statement)
“It is impossible to construct a device (operating in a cycle) that can transfer heat from cold body to the hot body without absorbing any work.”
Or
“Heat can not itself flow from colder body to a hotter body.”
3) Second law of thermodynamics based on entropy
“In all the spontaneous processes, the entropy of the universe increases.”
I hope you have clearly understood the definition of second law of thermodynamics.
If you have any queries, feel free to comment below.
Important Guides for you
Also read:
- What is thermodynamics? (35+ topics with definitions and examples)
- Zeroth law of thermodynamics
- What is First law of thermodynamics?
- First law of thermodynamics definition/statement (In simple way)
- Examples of First Law of Thermodynamics / Law of conservation of Energy
- First law of thermodynamics equation (A practical explanation)
- Limitations of First Law of Thermodynamics
- More about second law of thermodynamics
- Examples of Second Law of Thermodynamics (8+ best examples)
- Second Law of Thermodynamics Equation [Practical explanation]
- What is third law of thermodynamics? (in simple terms)
- Laws of thermodynamics
- What is Carnot Cycle in Thermodynamics?
- What is the definition of entropy in thermodynamics?
- Heat capacity vs specific heat in thermodynamics
- What is Thermodynamic Equilibrium? (With Best Example)
- Thermodynamic Process (With Examples)
- What is Thermodynamic System? – Open, Closed & Isolated system (With Examples)
