
Don’t worry, I will explain all these limitations of first law of thermodynamics along with the real life examples.
Well, before starting the limitations of first law of thermodynamics, you should know what the first law of thermodynamics is?
If you do not know anything about the first law of thermodynamics, kindly visit this article “detailed information on first law of thermodynamics.”
Hoping that you know all about 1st law, let’s get straight into its limitations.
Before that, just take a look at the statement of first law of thermodynamics.
First law of thermodynamics or law of conservation of energy:
“Energy can neither be created nor can be destroyed but transformation of one form into another form can be possible.”
Now let’s see the limitations.
The limitations of first law of thermodynamics are mentioned below.
1) First law does not say anything about whether the process is possible or not.
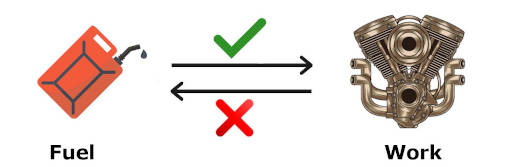
Let me explain to you with an example.
See the above image, you can see that in the engines, the chemical energy of the fuel is converted into useful mechanical work.
But we cannot convert this mechanical energy back into fuel. (Or in other words, engine cannot produce fuel back)
We all know that this is practically impossible. (Fuel can not be obtained back)
But the first law of thermodynamics does not says anything about it. Thus, this is one of the limitations of first law of thermodynamics.
2) First law of thermodynamics does not say whether the process will occur on its own or not
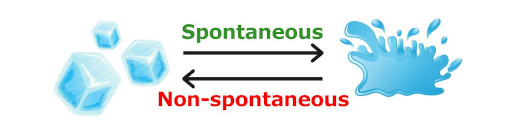
Don’t worry, I’ll explain.
Let me ask you a question. What will happen if you put ice on a table? It will melt down after some time on its own.
Now what about the reverse process (water to ice)?
Will this occur on its own?
The answer is NO.
We all know that conversion of water into ice does not occur on its own.
But, first law of thermodynamics does not say anything about this.
That means the first law of thermodynamics does not say whether this thermodynamic process occurs on its own or not. (In other words, first law of thermodynamics does not tell us whether the process is spontaneous or not)
3) First law does not provide any sufficient condition for a certain process to take place.
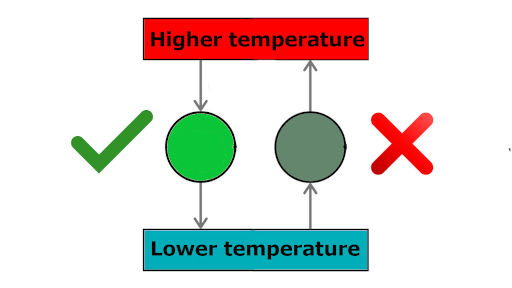
For example, you already know that heat always flows from higher temperature body to lower temperature body (see the green tick in above picture). But the reverse process is not possible (see wrong tick in above picture).
Now if we want to make this reverse process possible, then will it occur on its own?
Can we transfer heat from lower temperature body to higher temperature body without doing anything?
Is there any condition for transferring heat from lower to higher temperature body?
Do we need to supply anything to do this?
I don’t know. (Hahaha…)
Even the 1st law of thermodynamics does not say anything about this.
This is the limitation that it does not provide any condition for a process to occur.
What after these limitations?
Well, in order to solve all these limitations of first law of thermodynamics, the second law of thermodynamics was discovered.
You can check this second law of thermodynamics with 8+ amazing examples.
I Hope you have clearly understood the limitations of 1st law of thermodynamics. If you have any queries, feel free to comment below in the comment section.
Important Guides for you
Also read:
- What is thermodynamics? (35+ topics with definitions and examples)
- Zeroth law of thermodynamics
- What is First law of thermodynamics?
- First law of thermodynamics definition/statement (In simple way)
- Examples of First Law of Thermodynamics / Law of conservation of Energy
- First law of thermodynamics equation (A practical explanation)
- What is second law of thermodynamics? [8+ Best examples to remember the law]
- Examples of Second Law of Thermodynamics (8+ best examples)
- Second Law of Thermodynamics Definition/Statement (Next level explanation)
- Second Law of Thermodynamics Equation [Practical explanation]
- What is third law of thermodynamics? (in simple terms)
- Laws of thermodynamics
- What is Carnot Cycle in Thermodynamics?
- What is the definition of entropy in thermodynamics?
- Heat capacity vs specific heat in thermodynamics
- What is Thermodynamic Equilibrium? (With Best Example)
- Thermodynamic Process (With Examples)
- What is Thermodynamic System? – Open, Closed & Isolated system (With Examples)
