
Let me ask you a question.
In a scorching summer, what gets heats up faster on a sea shore?
A sand or a water?
The answer is “A sand”
But why does it happen like this?
The sand as well water receives the same amount of heat from the sun rays. Then why does sand gets heats up faster and water remains colder?
The reason is because of their heat capacity.
Yup, it’s a heat capacity.
Don’t worry, I’ll explain you everything about heat capacity and specific heat capacity with mind-blowing examples (scroll down to see images)
After reading this, You will get the exact conceptual knowledge as well as difference between the heat capacity and specific heat capacity along with real life examples.
So let’s get straight into it.
Contents
What is Heat capacity?
Definition
“Heat capacity (Hc): The ratio of heat supplied (Q) to the body to the change in it’s temperature ∆T is called heat capacity (Hc) of that body.”
Hc = Q/∆T
Or
“Heat capacity (Hc) is defined as the amount of heat absorbed (Q) in order to increase the temperature (∆T) by 1 unit.”
I know that you have not understood anything from this definition. (Hahaha)
Let’s get back to the examples of sand and water.
We saw in the above example that sand gets heated faster as compared to water.
From this, we can say that only few amount of heat is required to heat the sand. While a lot of energy is required to heat the water.
In other words, sand does not have more capacity to absorb the heat, while water keeps on absorbing the heat of the sun.

(Let me give you a funny example:
A short tempered person becomes angry very soon. While a calm person who keeps on tolerating everything will not become angry soon)
Heat capacity is also somewhat similar to this.
All the substances do not require the same amount of heat for increasing its temperature.
The amount of heat required to increase the temperature is different for different substances.
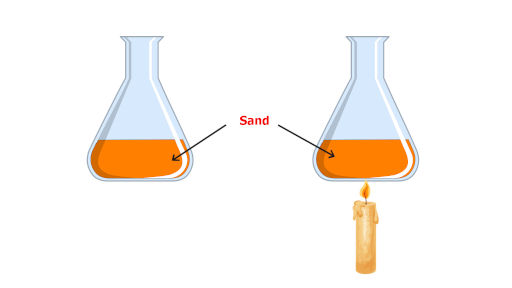
Now, I’ll show you this amazing example.
Let’s take an example of sand. As shown in the above picture, 1 kg of sand is filled in the flask.
Let us assume that initially the sand is at 20 °C. Now it is heated using a candle or a burner and its temperature reaches to 25 °C.
Let say, the amount of heat absorbed by the sand to increase its temperature from 20 °C to 25 °C is 1000 joules.
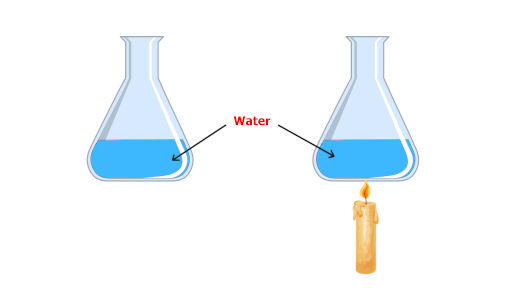
Now, we have to repeat the same procedure but the substance will be different here. The new substance is water.
The flask contains 1 kg of water initially at 20 °C. Let say we increase its temperature to 25 °C by using a burner or a candle.
Now the BIG QUESTION is how much heat will this water absorb?
Will it consume more heat or less heat as compared to that of sand?

Well, the answer is: Water will absorb more heat.
The quantity of sand and water is same. The temperature rise in sand and water is also same. But even though, the water absorbs more heat to reach the 25 °C temperature.
This is just because water has more capacity to absorb the heat. And this property is known as heat capacity.
Now let’s frame the proper definition. (Now, you will understand for sure)
Heat capacity (Hc): The ratio of heat supplied (Q) to the body to the change in it’s temperature ∆T is called heat capacity (Hc) of that body.
Hc = Q/∆T
Or
Heat capacity (Hc) is defined as the amount of heat absorbed (Q) in order to increase the temperature (∆T) by 1 unit.
[How to remember this?
Just see this formula, Hc = Q/∆T.
In this formula, if ∆T is 1, then Q = Hc. Means heat absorbed or rejected (Q) is known as Heat capacity (Hc)]
The unit of heat capacity is J/K.
Now, what did you notice from the above example of sand and water?
We noticed that the heat capacity of sand is less, while the heat capacity of water is more.
This clearly indicates that heat capacity depends on the material of the body.
Also, one more thing I would like to add. Heat capacity also depends on the mass of the object. Bigger the mass of the object, the more heat will be absorbed, so it’s heat capacity will be more. While smaller the mass of the object, less heat will be absorbed, so it’s heat capacity will be less.
I Hope you have clearly understood the definition of heat capacity.
Now, Let’s move to the next topic.
What is Specific heat capacity or specific heat (C)?
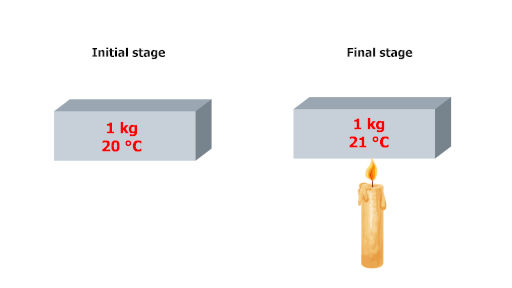
Now this is very simple.
You just have to take a unit mass over here. Rest of the things are same as that of heat capacity. That’s it!!
Don’t worry. I’ll explain.
In the above picture, you can see that the iron block has a mass of 1 kg. And it’s initial temperature is 20 °C.
After applying some heat, it’s temperature rises by 1 °C and finally it’s temperature becomes 21 °C.
During this phenomenon, the heat supplied is known as the specific heat capacity of that body.
Now let me give you a clear definition.
Specific heat capacity (C):
Specific heat capacity (C) is defined as the amount of heat absorbed (Q) in order to increase the temperature (∆T) by 1 unit, of a unit mass(m).
C = Q/m∆T
In other words, if the mass of a body is unity, then the heat capacity of the body is known as specific heat capacity or specific heat.
[How to remember?
Just see this formula C = Q/m∆T.
In this formula, if ∆T = 1 and m = 1, then Q = C. Means heat absorbed (Q) is known as Specific heat capacity (C)]
The unit of specific heat capacity is J/kg K.
Remember: The specific heat capacity of water is 4182 J/kg K.
That means 1 kg of water will absorb 4182 Joules of energy for raising its temperature by 1 K or 1 °C.
I hope you have clearly understood the definition of specific heat capacity. Now let me give you a difference between specific heat vs heat capacity.
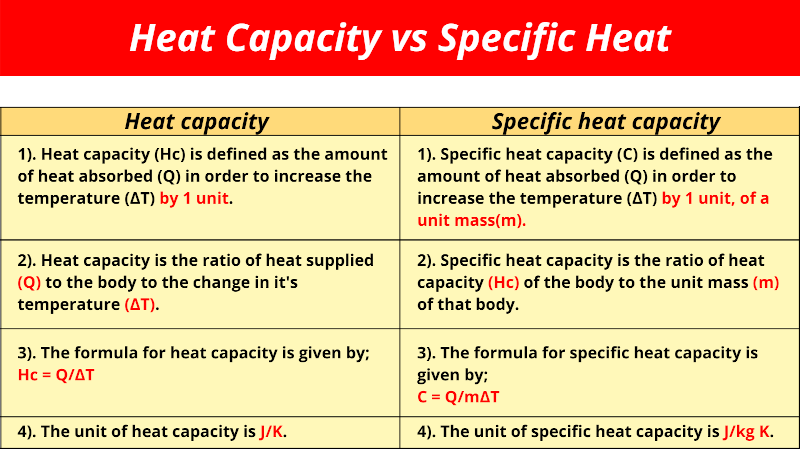
Heat capacity vs Specific heat capacity
| Heat capacity | Specific heat capacity |
| Heat capacity (Hc) is defined as the amount of heat absorbed (Q) in order to increase the temperature (∆T) by 1 unit. | Specific heat capacity (C) is defined as the amount of heat absorbed (Q) in order to increase the temperature (∆T) by 1 unit, of a unit mass(m). |
| Heat capacity is the ratio of heat supplied (Q) to the body to the change in it’s temperature ∆T. | Specific heat capacity is the ratio of heat capacity (Hc) of the body to the unit mass (m) of that body. |
| The formula for heat capacity is given by; Hc = Q/∆T | The formula for specific heat capacity is given by; C = Q/m∆T |
| The unit of heat capacity is J/K. | The unit of specific heat capacity is J/kg K. |
So this is it for Heat capacity vs Specific heat capacity.
I hope you have understood the concept as well as difference between heat capacity and specific heat capacity.
Well, I have also given you the image of the above differences which you can download from below.
If you have any doubts, feel free to comment in the comments section below.
Also let me know which example you liked the most.
Important Guides for you
Also read:
- What is thermodynamics? (35+ topics with definitions and examples)
- Zeroth law of thermodynamics
- What is First law of thermodynamics?
- First law of thermodynamics definition/statement (In simple way)
- Examples of First Law of Thermodynamics / Law of conservation of Energy
- First law of thermodynamics equation (A practical explanation)
- Limitations of First Law of Thermodynamics
- What is second law of thermodynamics? [8+ Best examples to remember the law]
- Examples of Second Law of Thermodynamics (8+ best examples)
- Second Law of Thermodynamics Definition/Statement (Next level explanation)
- Second Law of Thermodynamics Equation [Practical explanation]
- What is third law of thermodynamics? (in simple terms)
- Laws of thermodynamics
- What is Carnot Cycle in Thermodynamics?
- What is the definition of entropy in thermodynamics?
- What is Thermodynamic Equilibrium? (With Best Example)
- Thermodynamic Process (With Examples)
- What is Thermodynamic System? – Open, Closed & Isolated system (With Examples)
