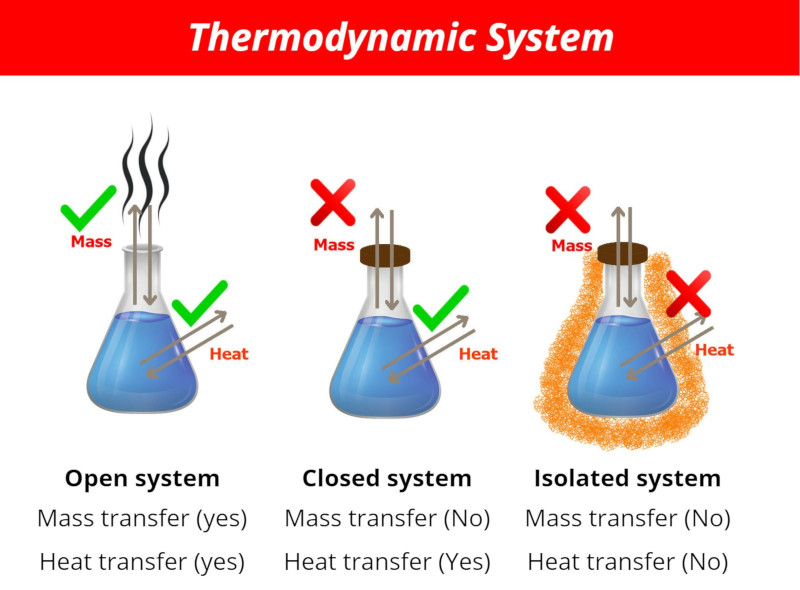
These are the Thermodynamic System.
- Open system
- Closed system and
- Isolated system
Let me tell you some basics before starting the types of Thermodynamic system.
What do you mean by thermodynamic system?
What do you mean by surrounding or environment?
What do you mean by boundary?
What is universe?
Here is the answer.
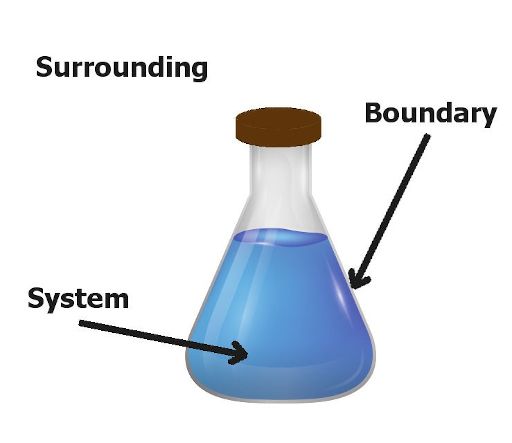
I’ll explain all these terms using this simple image.
The region which is inside the flask is known as a system.
The region which is outside the flask is known as surrounding.
And the imaginary line which separates the system and surrounding is known as a boundary.
Now, finally the universe is nothing but a combination of flask as well as its surroundings.
(It is similar like the stars, planets, satellites, etc combining to form a universe)
Now let me give you a proper definition of these terms.
Thermodynamic system: System is defined as a part of the universe which is chosen for thermodynamic study.
Surrounding: The region outside the system is known as surrounding.
Boundary: The actual or imaginary line which separates the system and surrounding is known as boundary (see figure).
Universe: The combination of system and the surroundings together is usually referred to as universe.
Yes this is simple!!!
Now you are done with the definitions of thermodynamic system, surrounding and universe.
Let’s proceed towards the types of thermodynamic system.
Contents
What are the types of thermodynamic system?
There are three main types of system.
1. Open system
2. Closed system
3. Isolated system
Open system
“The system in which heat transfer as well as mass transfer takes place with the surroundings is known as open system.”
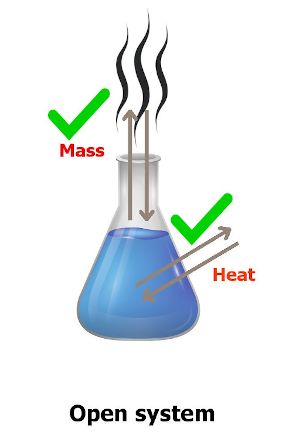
Have you understood anything from the definition and above image?
If not, don’t worry. I will explain to you in a simple way.
You can see in the above image that the transfer of mass of water takes place through the flask.
Why does this happen?
Because the flask is open. Right?
Also the heat transfer is taking place through this system (flask).
Thus a system in which mass transfer as well as heat transfer takes place with the surroundings is known as open system.
Closed system
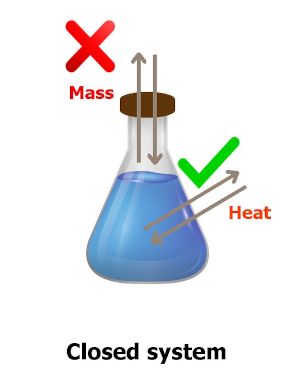
“The system in which only heat energy is exchanged with the surroundings is known as closed system.”
Now this is simple one.
If you have not understood anything from the image, read his below explanation.
You can see in the above image that the flask is closed.
As the flask is closed, there will be no mass transfer.
But, the heat can be transferred through the walls of the flask.
Thus the system in which only heat energy is exchanged with the surroundings is known as closed system.
Isolated system
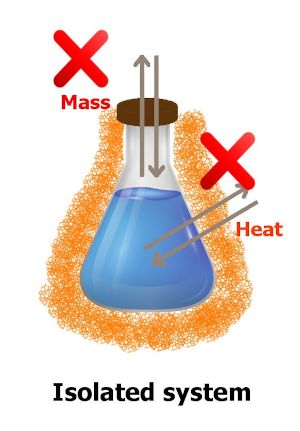
In this type of thermodynamic system, neither mass transfer takes place nor heat transfer takes place.
You can see in the above image that the flask is isolated using a non conducting material. Because of this, the heat transfer will not take place. Also the flask is closed, so there will be no mass transfer.
Such a system is known as isolated system.
You might have observed that the hot coffee remains hot and cold coffee remains cold in the thermos flask.
So the thermos flask is a very good example of isolated system.
Let me summarize the thermodynamic system in a single image and a table for better comparison.
Summary
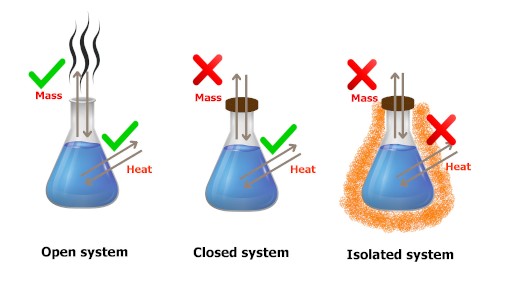
| Open system | Closed system | Isolated system |
| Mass transfer (yes) | Mass transfer (no) | Mass transfer (no) |
| Heat transfer (yes) | Heat transfer (yes) | Heat transfer (no) |
I hope you have understood the concept of thermodynamic system and surrounding. If you have any queries, feel free to comment below in the comment section.
Important Guides for you
Also read:
- What is thermodynamics? (35+ topics with definitions and examples)
- Zeroth law of thermodynamics
- What is First law of thermodynamics?
- First law of thermodynamics definition/statement (In simple way)
- Examples of First Law of Thermodynamics / Law of conservation of Energy
- First law of thermodynamics equation (A practical explanation)
- Limitations of First Law of Thermodynamics
- What is second law of thermodynamics? [8+ Best examples to remember the law]
- Examples of Second Law of Thermodynamics (8+ best examples)
- Second Law of Thermodynamics Definition/Statement (Next level explanation)
- Second Law of Thermodynamics Equation [Practical explanation]
- What is third law of thermodynamics? (in simple terms)
- Laws of thermodynamics
- What is Carnot Cycle in Thermodynamics?
- What is the definition of entropy in thermodynamics?
- Heat capacity vs specific heat in thermodynamics
- What is Thermodynamic Equilibrium? (With Best Example)
- Thermodynamic Process (With Examples)
