
What did you observe here?
Water always flows downward on its own.
But it never goes upward automatically.

What did you observe here?
A hot coffee becomes cold on it’s own. While it can not become hot again on its own.
Trust me. All the examples of second law of thermodynamics are very easy.
I’ll just show you the images like this and you are done with all the examples. (Scroll down to see more like this)
Well, let’s get started with some amazing real life examples of second law of thermodynamics.
But…But…But……
If you do not know anything about the second law of thermodynamics then I suggest you to refer this detailed article on the second law of thermodynamics which will make you understand all the three statements of the 2nd law.
I know you will not visit this above article. So let me share with you some glimpse of the statement of second law of thermodynamics here only.
Entropy statement of Second law of thermodynamics:
“In all the spontaneous processes, the entropy of the universe increases.”
Kelvin Planck’s statement:
“It is impossible to construct a device (operating in a cycle) which works on a single heat source and converts all of its heat completely into work”
Clausius’s statement:
“It is impossible to construct a device (operating in a cycle) that can transfer heat from cold body to the hot body without absorbing any work.”
Or
“Heat can not itself flow from colder body to a hotter body.”
If you are facing difficulty in understanding the above statements of second law, then kindly refer to this article “Detailed explanation on second law of thermodynamics“, where I’ve explained these statements in a very simple way.
Now let’s get straight into the examples of second law of thermodynamics.
Contents
Second law of thermodynamics examples
Examples based on Clausius’s statement
1) Air leaks from the balloon on its own

You might have noticed this on your birthday.
The air leaks from the balloon on its own after some time.
The air never goes inside the balloon on its own. This example is based on the Entropy statement of second law of thermodynamics. It is an example of spontaneous process.
2) Two gases will mix automatically on its own
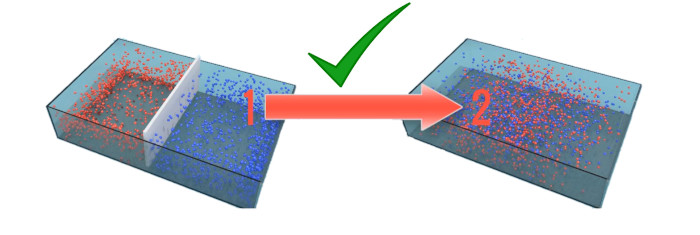
If we remove this white separator as shown in the image, then what happens?
Both the gases will get mixed with each other. And this process also occurs on its own. Thus this is an example of second law of thermodynamics which shows that the entropy of the universe increases due to this spontaneous process.
3) Hot coffee cools down automatically

This example is also based on the principle of increase in entropy.
In a shivering winter, if your mom prepares a hot coffee for you and you do not drink it within few minutes, then what happens to this coffee?
This coffee will cool down after some time. Right?
Yes. Obviously it will cool down. And this process occurs on its own (spontaneously).
As this process occurs spontaneously, the entropy of the universe will increase.
4) Object falls on the ground on its own

You have already experienced this.
A stone or any object always falls down on the ground on its own. These objects never goes up automatically.
This process of falling indicates that it is a spontaneous process and for such spontaneous processes, the entropy of the universe increases.
5) Ice melts automatically

You have already seen this at least once in your life.
What happens if you keep ice on a table for some time?
It starts melting.
Now this thermodynamic process occurs spontaneously (on its own).
Because of this spontaneous process, the entropy of the universe increases.
6) Water always flows from higher level to the lower level
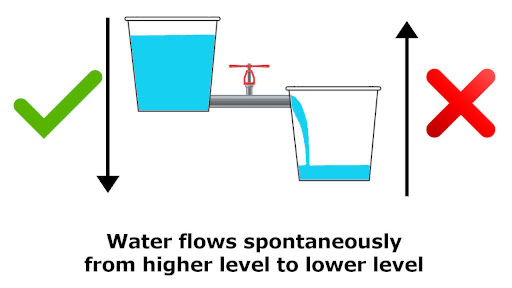
Have you seen water going upward automatically?
The answer is NO.
Because the water always flows spontaneously from higher level to the lower level.
It never goes up automatically.
Thus this spontaneous process of water flowing from higher level to lower level indicates the increase of entropy of the universe.
7) A gas takes the entire volume of the container
What will happen if you insert some gas in the closed container. Obviously it will spread throughout the container(on its own) and it will occupy the entire space of the container.
This is exactly the same as applying perfume on your shirt and the fragrance spreads in the entire room.
This process occurs spontaneously (i.e on its own) and because of this spontaneous process, the entropy of the universe increases.
Example based on Kelvin Planck’s statement
1) Cars and bikes engine

For understanding this example, you should know the Kelvin Planck’s statement of second law of thermodynamics.
Hope you know this Kelvin Planck’s statement, now let me explain to you how this example of car engine or bike engine is based on the 2nd law.
See, according to Kelvin Planck’s statement, there should be at least two thermal reservoirs (one at higher temperature and other at lower temperature) then only the engine will work.
In a car engine and bike engine, there is a higher temperature reservoir where heat is produced and a lower temperature reservoir where the heat is released.
Thus these engines are the example of second law of thermodynamics.
Example based on Clausius’s statement
1) Refrigerator using electricity to change the direction of heat flow

Before understanding this example, I suggest you to refer the Clausius’s statement of second law of thermodynamics.
You know what happens in the refrigerator?
The heat is traveling from the lower temperature body (i.e inside space of refrigerator) to the higher temperature body (i.e outside the refrigerator).
But this process is not possible on its own. To make this heat flow possible, there is a supply of external energy to this refrigerator. This external energy is nothing but electrical energy which is further used in the compressor of the refrigerator to produce mechanical work.
Thus this example satisfies the Clausius’s statement of second law of thermodynamics.
This is it for now.
Hope you have found this article helpful.
Let me know what you think about these examples of second law of thermodynamics. Feel free to comment if you have any queries.
Important Guides for you
Also read:
- What is thermodynamics? (35+ topics with definitions and examples)
- Zeroth law of thermodynamics
- What is First law of thermodynamics?
- First law of thermodynamics definition/statement (In simple way)
- Examples of First Law of Thermodynamics / Law of conservation of Energy
- First law of thermodynamics equation (A practical explanation)
- Limitations of First Law of Thermodynamics
- More topics on Second law of thermodynamics
- Second Law of Thermodynamics Definition/Statement (Next level explanation)
- Second Law of Thermodynamics Equation [Practical explanation]
- What is third law of thermodynamics? (in simple terms)
- Laws of thermodynamics
- What is Carnot Cycle in Thermodynamics?
- What is the definition of entropy in thermodynamics?
- Heat capacity vs specific heat in thermodynamics
- What is Thermodynamic Equilibrium? (With Best Example)
- Thermodynamic Process (With Examples)
- What is Thermodynamic System? – Open, Closed & Isolated system (With Examples)
