
The equation of Zeroth law of thermodynamics is mentioned below:
“If Ta = Tc and Tb = Tc, then according to zeroth law of thermodynamics Ta = Tb. Where Ta, Tb and Tc are the temperatures of body A, B and C respectively.”
Explanation
Consider three blocks A, B and C at different temperatures.
Place these blocks such that block A and block B are not in contact with each other.

Block A is in contact with block C, and in the same way block B is also in contact with block C.
After sometime, block A will be in thermal equilibrium with block C and similarly block B will also be in thermal equilibrium with block C.
Now if we keep block A and block B in contact with each other, then there won’t be any heat transfer between them.
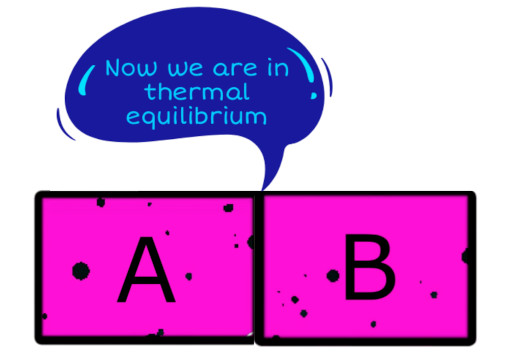
In other words, block A and block B are also in thermal equilibrium now.
This phenomenon is represented using the mathematical formula given below;
If Ta = Tc and Tb = Tc, then according to zeroth law of thermodynamics Ta = Tb. Where Ta, Tb and Tc are the temperatures of body A, B and C respectively.
I hope now you have understood the Zeroth law of thermodynamics equation.
If you want to see the detailed information about Zeroth law of thermodynamics, then visit the ultimate guide on Zeroth law of thermodynamics.
(This guide has all the important knowledge about Zeroth law of thermodynamics along with real life examples and lots more. You will love this guide for sure.)
Also see:
Suggested important topics:
- What is Thermodynamics? (35+ topics with Definitions and basics)
- What is First law of thermodynamics?
- First law of thermodynamics definition/statement (In simple way)
- Examples of First Law of Thermodynamics / Law of conservation of Energy
- First law of thermodynamics equation (A practical explanation)
- Limitations of First Law of Thermodynamics
- What is second law of thermodynamics? [8+ Best examples to remember the law]
- Examples of Second Law of Thermodynamics (8+ best examples)
- Second Law of Thermodynamics Definition/Statement (Next level explanation)
- Second Law of Thermodynamics Equation [Practical explanation]
- What is Third law of thermodynamics? (in simple terms)
- Laws of thermodynamics
- What is Carnot Cycle in Thermodynamics?
- What is the definition of entropy in thermodynamics?
- Heat capacity vs specific heat in thermodynamics
- What is Thermodynamic Equilibrium? (With Best Example)
- Thermodynamic Process (With Examples)
- What is Thermodynamic System? – Open, Closed & Isolated system (With Examples)
