
Yes, you read it right. It’s really really simple.
The second law of thermodynamics equation is mentioned below:

I know this equation may seems difficult to understand, but don’t worry I’m here to explain you the entire Equation of Second Law of Thermodynamics.
Here is the statement for this equation:
“In all the spontaneous processes, the entropy of the universe increases.”
This statement of second law of Thermodynamics is very important as it tells us whether the process will occur on it’s own or not.
If we can calculate the entropy change ∆S, then we can easily find out whether the process will occur on it’s own or not.
Don’t worry, I’ll explain you everything about this 2nd law equation using a simple example and I’ll also show you mathematical proof for the same.
You will surely come to know how this entropy equation (∆Suniverse >0) is related to the second law of thermodynamics.
Let’s get started.
Before that, I want to explain the term Entropy very quickly.
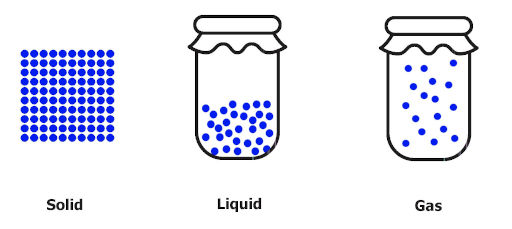
Just look at this image. What did you notice?
- The solids do not show any movement of molecules in it.
- The liquids shows more molecular motion as compared to solids, and
- The gases shows maximum movement of molecules.
Now, this movement of molecules is known as disorder or randomness of the molecules.
And entropy is nothing but the measurement of this disorder.
The equation of entropy is;
∆S = ∆Q/T
Now, I want to tell you one fact.
Whatever spontaneous (automatic) process occurs around us, are based on the second law of thermodynamics.
And all these spontaneous processes occurring around us satisfies this equation ∆Suniverse >0.
But how?
Let me give you an example, and here I’ll practically explain the second law of thermodynamics equation (with proof)

Take a hot cup of coffee on a table. Keep it as it is for few minutes. What will happen?
It will cool down after some time. Right? (In other words, coffee will reject heat to the surroundings)
Yes, it will. And we all know that.
But the big question is, what is the role of entropy in this process?
Well, entropy plays a very important role here.
Let’s assume the temperature of this coffee and temperature of surroundings.
Let;
-The temperature of coffee is 50 °C. (i.e 323 K)
-The temperature of surrounding is 20 °C. (i.e 293 K)
Now, there are two possible case
- The coffee may absorb heat from the surrounding, or
- The coffee may release heat to the surrounding.
But what is going to happen here in our case?
Will the coffee absorb the heat or will it release the heat?
Let’s find it out using the equation of second law of thermodynamics (∆Suniverse >0)
(Note: Actually you know that the coffee is going to lose heat to the surroundings, but there are many chemical reactions which we can not predict whether it will occur in this direction or that direction. So for such cases we can find this out using the equation of second law of thermodynamics ∆Suniverse >0)
Well, let’s see our case now. We have to see whether coffee will absorb heat from the surrounding or it will release heat to the surrounding.
Case 1: Coffee is absorbing heat from the surrounding
We have to check whether this thermodynamic process will occur on it’s own or not?
Let say coffee (system) absorbs 10 joules of heat from the surrounding.
That means, the surrounding will lose 10 joules of heat.
So we can write,
Qsystem = +10 J
Qsurrounding = -10 J
Now according to second law of thermodynamics, change in entropy of universe is given by the equation;
∆Suniverse = ∆Ssystem + ∆Ssurrounding
= ∆Qsystem/Tsystem + ∆Qsurrounding/Tsurrounding
= (+10/ 323) + (-10/293)
= -0.00316 J/K
This is a negative value,
The entropy of the universe is decreasing here. Thus it violates the equation of second law of thermodynamics (∆Suniverse should be greater than 0, but here the answer is negative).
So, we can say that this process is non spontaneous. It will not occur on its own. This process of absorbing heat from the surrounding will not occur on its own.
Now, let’s take another case.
Case 2: Coffee is releasing heat to the surrounding
We have to check whether coffee will release heat automatically or not?
Let say coffee (system) releases 10 joules of heat to the surrounding.
That means, the surroundings will absorb 10 joules of heat.
So we can write,
Qsystem = -10 J
Qsurrounding = +10 J
Now, change in entropy of universe is given by equation;
∆Suniverse = ∆Ssystem + ∆Ssurrounding
= ∆Qsystem/Tsystem + ∆Qsurrounding/Tsurrounding
= (-10/ 323) + (+10/293)
= +0.00316 J/K
This is a positive value,
Kudos!!
The entropy of the universe is increasing here. Thus it satisfies the equation of second law of thermodynamics (∆Suniverse should be greater than 0).
So, we can say that this process is spontaneous. This process of releasing heat to the surrounding will occur on its own.
Well, in the above example you were already knowing that coffee is going to lose heat to the surrounding.
But in some other examples of chemical reactions, you may not be able to predict whether the chemical reaction will occur on its own or not.
So, by calculating the entropy change for that particular reaction, we can easily decide whether the reaction will occur on it’s own or not.
I hope you have clearly understood the equation of Second law of thermodynamics with a practical example.
If you have any doubts, feel free to comment below in the comments section.
It makes me feel good after helping you.
Important Guides for you
Also read:
- What is thermodynamics? (35+ topics with definitions and examples)
- Zeroth law of thermodynamics
- What is First law of thermodynamics?
- First law of thermodynamics definition/statement (In simple way)
- Examples of First Law of Thermodynamics / Law of conservation of Energy
- First law of thermodynamics equation (A practical explanation)
- Limitations of First Law of Thermodynamics
- What is second law of thermodynamics? [8+ Best examples to remember the law]
- Examples of Second Law of Thermodynamics (8+ best examples)
- Second Law of Thermodynamics Definition/Statement (Next level explanation)
- What is third law of thermodynamics? (in simple terms)
- Laws of thermodynamics
- What is Carnot Cycle in Thermodynamics?
- What is the definition of entropy in thermodynamics?
- Heat capacity vs specific heat in thermodynamics
- What is Thermodynamic Equilibrium? (With Best Example)
- Thermodynamic Process (With Examples)
- What is Thermodynamic System? – Open, Closed & Isolated system (With Examples)
