In physics, the second law of thermodynamics deals with two devices;
- Heat engine and
- Heat pump (or refrigerator).
The second law of thermodynamics for the heat engine is known as Kelvin-Plank’s statement.
While the second law of thermodynamics for heat pump is known as Clausius statement.
Let’s discuss these two statements below.
Kelvin Planck’s statement for heat engine
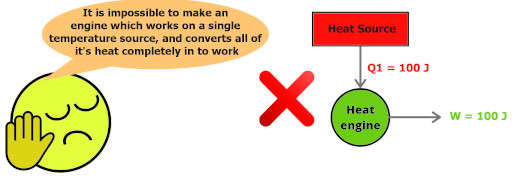
Kelvin Planck’s statement:
“It is impossible to construct a device (operating in a cycle) which works on a single heat source and converts all of its heat completely in to work”
I know it’s difficult to understand such statements given by great physicists.
So simply remember the below point.
This Kelvin Planck’s statement simply wants to say that – “A heat engine must exchange the heat with at least two thermal reservoirs, one at higher temperature and other at lower temperature, then only the engine will operate.”
Or
“Perfect engine with 100% efficiency is impossible”
This is only the simple meaning of the Kelvin-Plank’s statement.
Now let us see Clausius statement for second law of thermodynamics.
Clausius statement for heat pump
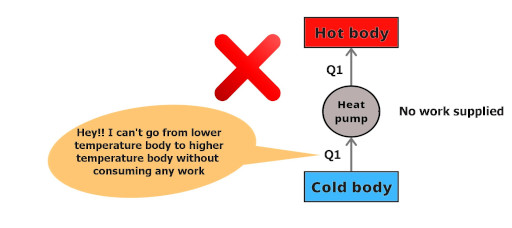
Clausius’s statement:
“It is impossible to construct a device (operating in a cycle) that can transfer heat from cold body to the hot body without absorbing any work.”
Or
“Heat can not itself flow from colder body to a hotter body.”
It’s very easy to understand, just see the above picture. Heat Q1 is at a lower temperature body and it is saying that it will not travel to a higher temperature body without absorbing any heat.
I hope you have understood the statement of the second law of thermodynamics in physics.
For more detailed information on Second law of thermodynamics, check the ultimate guide on Second law of thermodynamics.
(This guide has all the important knowledge about Second law of thermodynamics along with real life examples and lots more. You will love this guide for sure.)
Also see:
Suggested important topics:
- What is Thermodynamics? (35+ topics with Definitions and basics)
- What is First law of thermodynamics?
- First law of thermodynamics definition/statement (In simple way)
- Examples of First Law of Thermodynamics / Law of conservation of Energy
- First law of thermodynamics equation (A practical explanation)
- Limitations of First Law of Thermodynamics
- What is second law of thermodynamics? [8+ Best examples to remember the law]
- Examples of Second Law of Thermodynamics (8+ best examples)
- Second Law of Thermodynamics Definition/Statement (Next level explanation)
- Second Law of Thermodynamics Equation [Practical explanation]
- What is Third law of thermodynamics? (in simple terms)
- Laws of thermodynamics
- What is Carnot Cycle in Thermodynamics?
- What is the definition of entropy in thermodynamics?
- Heat capacity vs specific heat in thermodynamics
- What is Thermodynamic Equilibrium? (With Best Example)
- Thermodynamic Process (With Examples)
- What is Thermodynamic System? – Open, Closed & Isolated system (With Examples)
