Here you will get the exact reason how first law of thermodynamics is applied to the photosynthesis process.
First of all, first law of thermodynamics states that;
“Energy can neither be created nor it can be destroyed. But it can be transformed from one form to another.”
We all know that we need a cooking stove, oven, etc to prepare our food.
Similarly the plants also require sunlight to prepare their food.
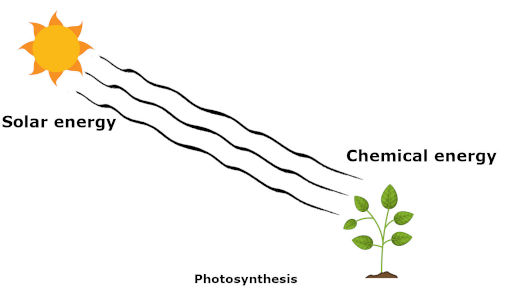
Now in a photosynthesis process, the solar energy is received by the plants during the day time.
The solar energy of the sun is absorbed by the plants and it is converted into molecular potential energy in the form of sugar (which is known as a chemical energy).
In short, during the photosynthesis process, the solar energy or light energy of the sun is converted into chemical energy of plants.
Thus plants are not generating their own chemical energy, but this chemical energy is available from the conversion of solar energy.
Finally, during the photosynthesis process, according to the first law of thermodynamics the energy remains conserved.
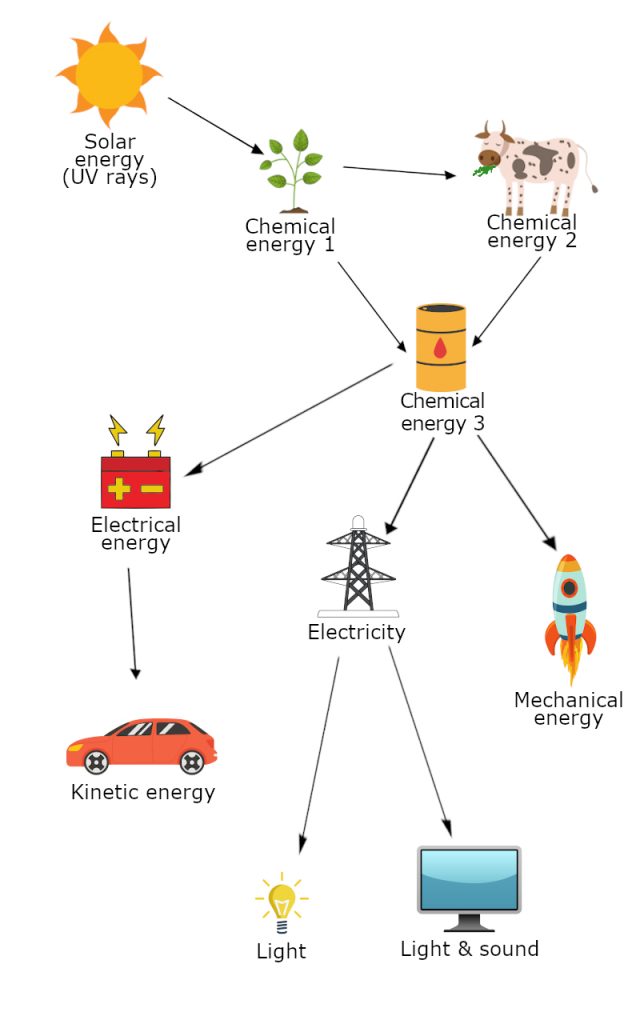
You can see how chemical energy of the plants gets further converted into other energies.
For more detailed information on First law of thermodynamics, check the ultimate guide on First law of thermodynamics.
(This guide has all the important knowledge about First law of thermodynamics along with real life examples and lots more. You will love this guide for sure.)
Also see:
Suggested important topics:
- What is Thermodynamics? (35+ topics with Definitions and basics)
- What is First law of thermodynamics?
- First law of thermodynamics definition/statement (In simple way)
- Examples of First Law of Thermodynamics / Law of conservation of Energy
- First law of thermodynamics equation (A practical explanation)
- Limitations of First Law of Thermodynamics
- What is second law of thermodynamics? [8+ Best examples to remember the law]
- Examples of Second Law of Thermodynamics (8+ best examples)
- Second Law of Thermodynamics Definition/Statement (Next level explanation)
- Second Law of Thermodynamics Equation [Practical explanation]
- What is Third law of thermodynamics? (in simple terms)
- Laws of thermodynamics
- What is Carnot Cycle in Thermodynamics?
- What is the definition of entropy in thermodynamics?
- Heat capacity vs specific heat in thermodynamics
- What is Thermodynamic Equilibrium? (With Best Example)
- Thermodynamic Process (With Examples)
- What is Thermodynamic System? – Open, Closed & Isolated system (With Examples)
